
Meet the tCOM+
Developed with care, designed for confidence.
The tCOM+ offers continuous monitoring of tcPCO2, SpO2, and PR with a sleek new look and feel. This latest model includes significant software advances that improve workflow for providers, while continuing to support reduced blood draws and proactive ventilator management across care areas.
Accurate
Continuous, noninvasive CO2 values tightly correlate with PaCO2 regardless of ventilation strategy or degree of V/Q mismatch.

Easy
Long site times, a range of attachment solutions, and sensor features like Smart Cal-Mem mean smoother monitoring experiences at the bedside.
Safe
Low temperatures and design features promoting skin integrity support safe monitoring in even the smallest, most fragile patients.
Trusted
Recommended in a variety of clinical practice guidelines by respected organizations all over the world.
Monitoring that does more.
More efficiency:
- Resolve issues quickly with helpful alerts and on-screen tutorials.
- Proactively manage and maintain equipment with dedicated sensor information screens.
- Keep bedside and equipment areas organized with integrated cable management.
- Smart Cal-Mem feature allows temporary disconnection for bathroom breaks, repositioning, and other workflows.

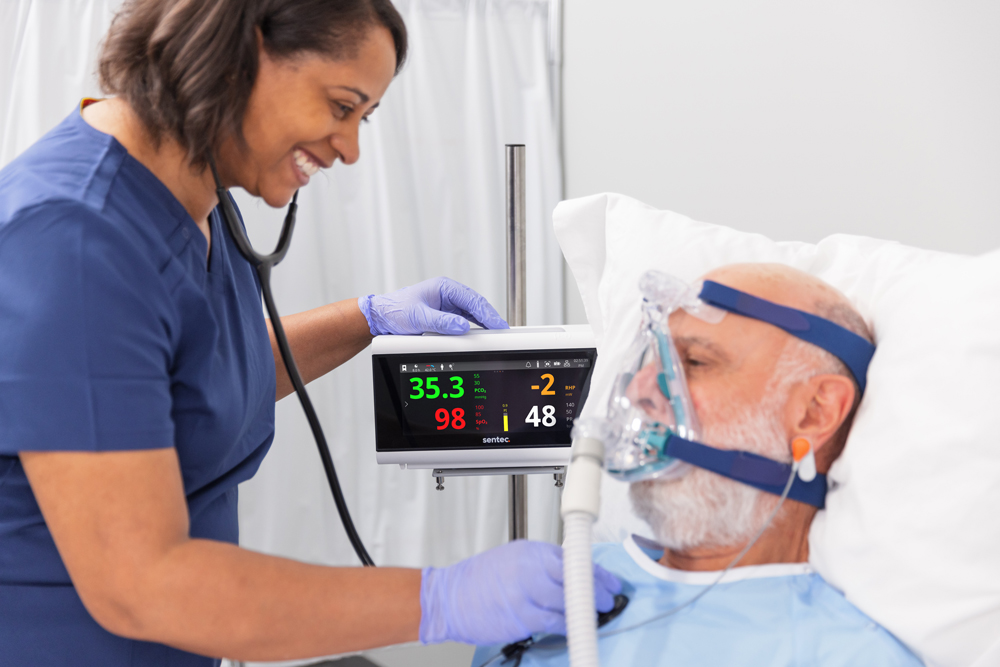
More flexibility.
- Carry the lightweight tCOM+ by its fold-flat handle or choose a mounting solution that fits your team’s needs.
- Connect with patient monitoring systems (PMS), patient data management systems (PDMS), and polysomnographic systems (PG/PSG).
- Compatible with the V-SignTM sensor
Looking for support?
Visit this link and select a product to browse relevant support resources.
Get info
Looking for a little more info on one of our products?
Let us know.
Book a Meeting
We're always available to discuss our products & work out a plan with you.
Arrange a Demo
We are more than happy to arrange a demo session to showcase our products.
Ask a Question?
The team at Apex Medical can answer your questions via phone or email.
Contact Us
Reach out and contact with any general questions or enquiries.
